Pyrene azide 2
| Cat. # | Quantity | Price | Lead time | Buy this product |
|---|---|---|---|---|
| A2530 | 1 mg |
$110
|
in stock | |
| B2530 | 5 mg |
$210
|
in stock | |
| C2530 | 10 mg |
$310
|
in stock | |
| D2530 | 25 mg |
$410
|
in stock | |
| E2530 | 50 mg |
$695
|
in stock |

This pyrene azide is a derivative of pyreneacetic acid with triethyleneglycol (PEG3) linker.
An alternative pyrene azide with a shorter linker is also available. A detailed description of the fluorophore can be found there.
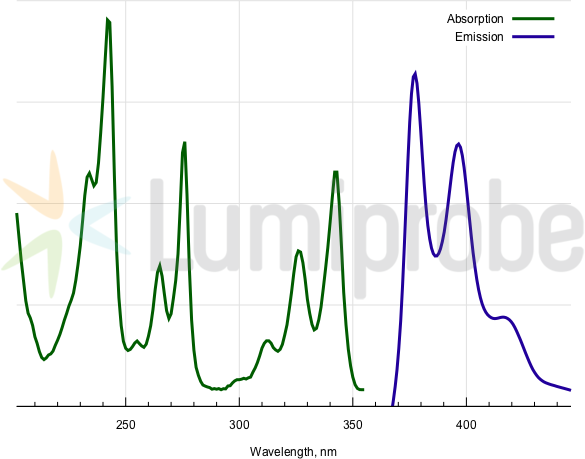
Absorption and emission spectra of pyrene fluorophore
Recommended protocol
Calculator
Customers also purchased with this product
General properties
| Appearance: | yellowish solid |
| Molecular weight: | 416.47 |
| CAS number: | 1807512-45-3 |
| Molecular formula: | C24H24N4O3 |
| Solubility: | good solubility in chloroform, dichloromethane, moderate solubility in DMSO, DMF, acetonitrile |
| Quality control: | NMR 1H (95%) |
| Storage conditions: | Storage: 24 months after receival at -20°C in the dark. Transportation: at room temperature for up to 3 weeks. Avoid prolonged exposure to light. |
| MSDS: | Download |
| Product specifications |
Spectral properties
| Excitation/absorption maximum, nm: | 343; 326; 313; 276; 265; 242; 234 |
| Emission maximum, nm: | 377; 397 |
Product citations
- Bednarczyk, M.; Peters, J.K.; Kasprzyk, R.; Starek, J.; Warminski, M.; Spiewla, T.; Mugridge, J.S.; Gross, J.D.; Jemielity, J.; Kowalska, J. Fluorescence-Based Activity Screening Assay Reveals Small Molecule Inhibitors of Vaccinia Virus mRNA Decapping Enzyme D9. ACS Chemical Biology, 2022, 17(6), 1460-1471. doi: 10.1021/acschembio.2c00049
- Wanat, P.; Walczak, S.; Wojtczak, B.A.; Nowakowska, M.; Jemielity, J.; Kowalska, J. Ethynyl, 2-Propynyl, and 3-Butynyl C-Phosphonate Analogues of Nucleoside Di- and Triphosphates: Synthesis and Reactivity in CuAAC. Organic Letters, 2015, 17(12), 3062–3065. doi: 10.1021/acs.orglett.5b01346
This Product is offered and sold for research purposes only. It has not been tested for safety and efficacy in food, drug, medical device, cosmetic, commercial or any other use. Supply does not express or imply authorization to use for any other purpose, including, without limitation, in vitro diagnostic purposes, in the manufacture of food or pharmaceutical products, in medical devices or in cosmetic products.
Short link - lumiprobe.com/sh/p/1c
The count of items is incorrect.
























 $
$ 
